|
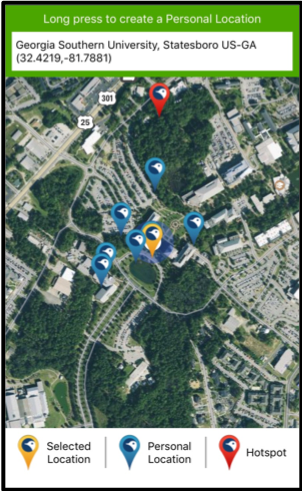
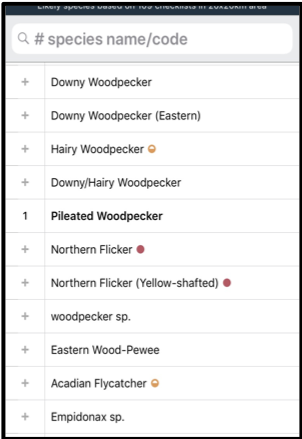
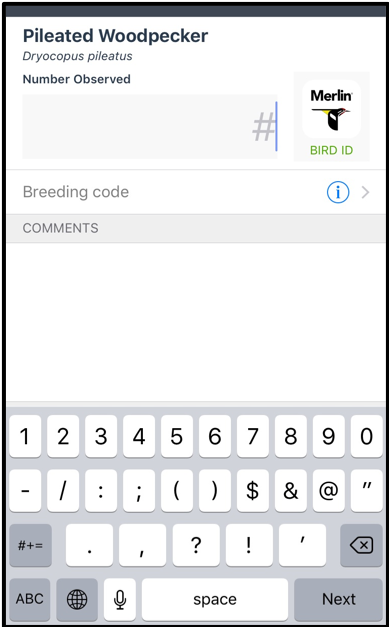
Birds: Using eBird to create a connection between kids and nature Written by Dr. Heather Scott, Instructor of Science Education at Georgia Southern University Picture this: It is February 16th, 5:30 am and five kids head out onto a nature trail to look for owls! Sounds unbelievable? Well, it's true. Our family rented a cabin in the Okefenokee Swamp to bird watch, and our teenagers spent every free moment exploring outside to add birds to their eBird lists. Now I may be mistaken, but that’s not typically how teenagers are spending their time on their phones these days. Yet, these kids have all created accounts and are adding to their life lists as often as they can. If you are even mildly interested in birds and want to learn more and contribute to a worldwide database then eBird is the place to start. So, how DO you get started?Your first step is to create an account before you can enter your sightings. Once you register for an account, download the eBird app to use on your phone and use your login credentials. Now that you have set up an eBird account, then you are ready to record observations.
What are you waiting for?
Birds are outside right now, waiting for you to notice them! You don’t have to plan an elaborate trip or even leave your neighborhood. You can observe the birds right outside your window to start your bird list on eBird. The bird photos included in this blog were taken by my daughter, Emmaline Scott, on the trip to the swamp. Her fascination with birds has extended to nature photography.
0 Comments
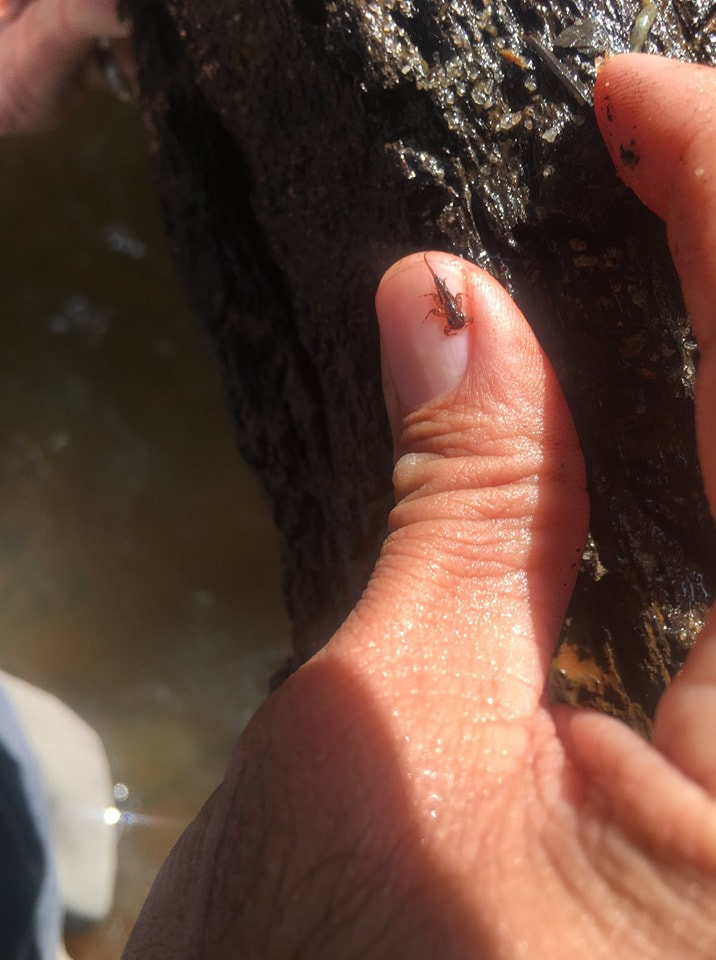
WHY BENTHIC MACROINVERTEBRATES?
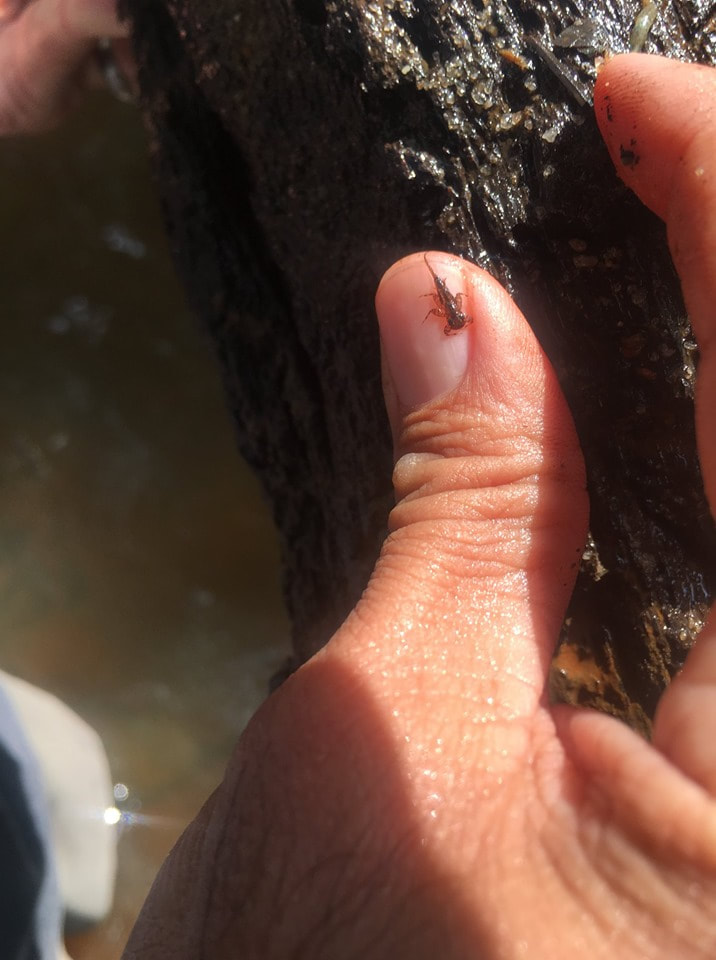
Written by Dr. Checo Colon-Gaud, Associate Professor of Biology at Georgia Southern University If memory serves me right, I’ve spent most of my life around rivers and streams. Stumping across creeks and flipping rocks as a kid to fishing and paddling as I grew older. However, it wasn’t until I was in my twenties that I truly appreciated the great diversity of organisms that lived beneath the water’s surface. Little by little I became more intrigued by a particular group of creatures that appeared to rule these underwater habitats. Benthic macroinvertebrates. The word benthic (or benthos) refers to those organisms that live in the bottom substrates and macroinvertebrate are organisms that lack a backbone and usually are large enough to see with the naked eye. Well…usually. These bottom-dwelling invertebrates can be vastly abundant in freshwater systems (rivers, lakes, wetlands) and can often be dominated by immature, larval, or nymph stages of aquatic insects. Aquatic insects can be incredibly diverse in freshwater systems and they tend to be ubiquitous to almost all aquatic environments. They can also be relatively long-lived in aquatic environments, with some spending over a year of their life cycles as immature stages, whereas their terrestrial adult stages can often only last several hours to several days. This unique life history of shared aquatic and terrestrial life stages makes them important conduits of energy between the water and the land, as many adults return to the water to deposit energy-rich eggs and young that provide a food source to many fish and other aquatic consumers. Whereas those that survive to became aerial adults often become a prey source to terrestrial consumers such as birds, spiders, etc. Aquatic insects are not only taxonomically diverse, but also functionally diverse. Meaning that they can play important roles during their aquatic life stages that support essential ecosystem services such as the decomposition of organic matter and the cycling of energy and nutrients. Many of these ecosystem services can be associated with the maintenance of healthy aquatic environments and clean water. And again, let’s not forget that they are also an important food source to fish, waterfowl, and other valuable aquatic organisms. Aquatic insects and other benthic macroinvertebrates are also important indicators of ecosystem health and condition. Because of their high levels of diversity and unique adaptations to endure life in water, many benthic invertebrates are known to be sensitive to pollution and other alterations to aquatic ecosystems. The presence of high numbers (i.e., abundance) or high levels of diversity of sensitive groups of benthic invertebrates can usually be associated with ideal water quality conditions such as high dissolved oxygen content. On the other hand, freshwater systems that are mostly dominated by somewhat tolerant to highly tolerant taxa of benthic invertebrates can be often associated with alterations or impairment to the system (e.g., pollution, habitat loss, etc.). Freshwater scientists have used benthic macroinvertebrates as a tool for assessing the health of freshwater systems for decades as these can provide a more in depth look at the conditions of the system in real time. The basis for this is that the organisms that are present in the system are exposed to such conditions continuously. Benthic macroinvertebrates can teach us a lot about the systems they inhabit. Not only are they essential role players in the functioning of freshwater ecosystems, but they help sustain the organisms that we most often associate with these systems. Gaining an appreciation of benthic macroinvertebrates, as well as the diversity and function of the aquatic organisms that live underneath the water can provide a valuable way to protect and conserve these important natural resources. What is Environmental Chemistry?
Written by Dr. Shainaz Landge, Assistant Professor of Chemistry at Georgia Southern University Environmental Science is a multidisciplinary field that can include chemical, physical and biological sciences working together to study and find effective solutions for environmental problems.1 There are multiple branches of environmental sciences2
Common quality measurements targeting environmental chemistry include Dissolved Oxygen (DO), pH, temperature, conductivity, clarity and salinity. In water quality monitoring, DO is essential for aquatic organisms, plants, and bacteria to survive. It is measured in parts per million (ppm) or mg/L. The water temperature has a strong influence (inverse relationship) on DO. At lower temperatures, there are higher dissolved oxygen levels.4 Fluctuations in dissolved oxygen can impact the health of a water way by disrupting plant growth, macroinvertebrates, and bacteria. To measure dissolved oxygen environmental chemists needs to fix the water sample and use titration study. The pH test (acidity measure) is one of the most critical indicators for water health. It measures the Hydrogen ion (H+) concentration in the water. Generally, solutions below 7.0 are acidic in nature and above 7.0 are basic. pH can be influenced by a variety of conditions including both man-made (agriculture) and environmental (hurricanes). To meet the Georgia state standards, the pH should be in the range of 6.0 to 8.5. Temperature is an important factor for aquatic life, and state standards suggest that it should be less than 32.2 oC (90 oF). Small, measured, seasonal changes are manageable by aquatic species, but a sudden change of degree or two can create shock or stress in aquatic species. The cold-shock,5 which results from the rapid decrease in water temperature, can bring number of physiological and behavioral changes in species, which can lead to death. In order to get accurate readings on other chemical reactions in water, chemists must know what is happening to the water temperature first. Conductivity is the measure of the ability of water to pass an electric current. It can be affected by the amounts of nutrients or ions present in the water. Conductivity is easily measured by chemists by analyzing the concentrations of different ions in the water, however this can be time consuming so most often use conductivity probes /meters. Temperature significantly affects conductivity. Higher temperatures will have higher conductivity levels, as it will help to dissociate the ions present in the water. Common ions that conduct electrical currents in water are sodium, chloride, calcium, and magnesium. Salinity is the concentration of dissolved salts in water, and it severely affects the plant and animal life. This particular study is important when dealing with ocean water, where the salinity of average ocean water is 35 parts per thousand (ppt). Salinity affects conductivity as well and will increase with higher levels of salt in the water. Because aquatic species are adapted to certain levels of salinity, changes can adversely affect them. Clarity, as the name suggests, is the clearness of the water and is affected by the suspended particles and algae in the water. Water with lower clarity will limit the amount of sunlight needed for photosynthesis resulting in the potential of suffocation of aquatic organisms. Clarity is usually measured with the naked eye and on a scale. Other measurements, which can be studied, are presence of nutrients (nitrates and phosphates) and heavy metal contamination.6 Nutrients such as nitrates are found in the water from fertilizers or animals waste (sewage) and phosphates are generally found in water from soaps, fertilizer etc. An excess of nutrients can lead to environmental impacts such as algal blooms and decreased dissolved oxygen levels. Heavy metal contamination from lead, zinc, cadmium, or mercury can occur through mining, manufacturing or use of synthetic products such as pesticides, paints, industrial waste, or domestic sludge in water bodies.7 Other quantitative research measurements can be conducted by instruments such as Gas Chromatography-Mass Spectrometry (GC-MS), High Performance Liquid Chromatography (HPLC), Atomic Absorption Spectra (AAS), and Inductively Coupled Plasma - Mass Spectrometry (ICP-MS). These are generally used to detect metal ion concentrations as well as anion and cation concentrations and their distributions.8 While we often think of water health as a biological construct, it is easy to see why chemists are invaluable partners when it comes to monitoring and studying water ways. It is not just important to know how to collect the data but also the process and what the outcomes mean in terms of overall health, causes of rising or falling chemical fluctuations, and how to conduct further testing to nuisance out potential issues. By studying the chemical reactions in the environment, chemists contribute to the monitoring of our waterways. References:
|
Details
Archives
June 2019
CategoriesSupported by the Gulf Research Program of the National Academies of Sciences, Engineering, and Medicine under the Grant Agreement: 20000G9576
|
OUR2SWAMP








 RSS Feed
RSS Feed
